Drug development for metabolic dysfunction-associated steatohepatitis (MASH) has long been constrained by heavy reliance on invasive and costly liver biopsy. FDA’s acceptance of a Letter of Intent (LOI) to evaluate liver stiffness measurement (LSM) by vibration-controlled transient elastography (VCTE) as a reasonably likely surrogate endpoint in clinical trials marks an important regulatory signal for non-invasive test in MASH. Here, TriApex’s team provides an end-to-end overview of VCTE in MASH,
*from clinical context that creates demand for non-invasive tool,
*to how VCTE is used in the real world,
*and how it is currently being evaluated within regulatory pathways.
We hope this overview could provide a practical perspective that helps peers in the field interpret where VCTE fits today.
★ Table of Contents ★
01Disease and Regulatory Background
02Clinical Maturity and Real-world Grounding of VCTE
03VCTE Measurements in Clinical Trials
04Regulatory Considerations and Current Use in Animals
05Conclusion
1.1 Why MASH is uniquely challenging?
Metabolic dysfunction-associated steatotic liver disease (MASLD) affects approximately 38% of the global adult population, with higher prevalence in certain subpopulations, including those with obesity and type 2 diabetes (T2D)[1]. The disease spans a continuum from steatosis to steatohepatitis, fibrosis, cirrhosis, and ultimately hepatocellular carcinoma. Liver fibrosis is commonly staged on a five-point scale (F0-F4), with increasing scale reflecting cumulative injury[2].
Figure 1 Disease spectrum of MASLD[3]
MASH represents the active form of MASLD. Patients with ≥F2 fibrosis are considered to have “at-risk MASH”, as fibrosis at this stage is a strong predictor of all-cause mortality and liver related outcomes and generally warrants medical intervention. Patients with F2-F3 fibrosis have become the primary focus of current clinical programs[2].
Effective drug development for MASH remains challenging due to the heterogeneous nature of the disease, slow and variable progression and the central role of fibrosis in long-term outcomes[4]. These factors complicate patient stratification, endpoint selection and demonstration of benefit in clinical trials, creating a gap between regulatory expectations and practicable endpoints.
1.2 Liver biopsy as the gold standard
Liver biopsy (LB) has long served as the histologic gold standard for diagnosing MASH and evaluating therapeutic efficacy. It remains the only approach to provide direct liver microscopic assessment of key histological features in MASH, including steatosis, lobular inflammation, hepatocyte ballooning, and fibrosis architecture[5]. Given the impracticality of capturing definitive outcomes within feasible trial durations, the FDA has accepted histology-based surrogate endpoints for Phase 3 trials, including[6]:
* Histopathologic findings showed either resolution of steatohepatitis or improvement in fibrosis, without worsening of the other;
* Both
Figure 2 FDA approved surrogate endpoints for MASH phase 3 trials[6]
However, LB has well-recognized limitations. Its invasiveness, high cost, procedural risks, and inter- and intra-observer variability restrict its scalability and longitudinal follow-up[3][7]. The need for biopsy-proven fibrotic MASH causes 70-80% of screening failures in clinical trials since many patients do not have fibrotic MASH on the baseline[8].
These constraints have driven emerging interest in non-invasive tests (NITs) to support more accurate MASH pre-screening, diagnosis, and disease staging.
Figure 3 A landscape of non-invasive tests (NITs) used for MASLD[3]
1.3 FDA’s stated position on non-invasive biomarkers The FDA has signaled support for the use of non-invasive biomarkers in MASH drug development. Sponsors are encouraged to scientifically justified the incorporation of these biomarkers from proof-of-concept Phase 2 trials through late-stage trials[9]. Draft FDA guidance also emphasizes the evaluation of such biomarkers as potential surrogate endpoints, provided they demonstrate diagnostic and staging performance comparable to or better than LB[10].
Figure 4 FDA draft guidance on developing drugs for noncirrhotic nonalcoholic steatohepatitis[10]
In this context, FDA CDER’s acceptance of the LOI for Drug Development Tool (DDT) under the Biomarker Qualification Program in August 2025 from Echosens marks a milestone for the field. The proposed LSM by VCTE as a “reasonably likely surrogate endpoint” for clinical trials intends to predict the risk of all-cause mortality or liver-related events in MASH adults with moderate-to-several (F2-F3) fibrosis[11]. While qualification plan (QP) and full qualification package (FQP) are still required before final determination, this signals regulatory openness and could potentially establish a pathway for NITs in MASH drug development. Figure 5 Letter of Intent from FDA[11]
VCTE is widely used in routine liver care and has accumulated extensive real-world evidence. This provides a strong foundation for evaluating VCTE within regulatory framework, where credibility, reproducibility, and generalizability are essential. Broad real-world adoption helps address concerns about whether a method performs reliably outside of tightly controlled study settings.
2.1 What VCTE measures
VCTE estimates liver tissue stiffness by generating a shear wave and tracking its propagation using pulse-echo ultrasound. Stiffer liver tissue produces higher measured value. Most published VCTE evidence is derived from FibroScan devices, which have been used to assess liver fibrosis since 2007[12]. Measurements are highly reproducible, with excellent interobserver agreement[13]. VCTE provides two primary quantitative outputs[14]:
☆ Liver Stiffness Measurement (LSM)
Reported in kPa (Measurement range: 1.5 – 75 kPa)
Serves as a mechanical proxy for the degree of hepatic fibrosis
☆ Controlled Attenuation Parameter (CAP)
Reported in dB/m (Measurement range: 100 – 400 dB/m)
Correlates with the degree of hepatic steatosis
To improve diagnostic confidence, several composition scores have been developed that integrate routine serum biomarkers and clinical variables. Commonly used scores are summarized below[15][16].
Figure 6 Commonly used FibroScan-based scores in clinics[15][16]
VCTE supports MASLD care from initial assessment to longitudinal follow-up, with applications being grouped into three broad categories[14]:
☆ Phenotyping and risk stratification
VCTE aids in characterizing disease burden and identifying patients at risk who may benefit from specialist referral. Major international MASLD/NAFLD clinical guidelines incorporate VCTE within sequential risk-stratification pathway to support referral of patients in the target high-risk population (e.g., T2D, obesity and liver enzyme abnormalities)[5][17][18][19].
Guidelines typically adopt simplified, standardized cutoffs to support risk stratification in large, heterogeneous populations. The practical question most guidelines aim to answer is not “What exact fibrosis stage is this?”, but rather “Who is at low risk, and who warrants referral or further evaluation?”
Figure 7 Summary of guideline-recommended risk stratification and referral strategies
Across guidelines, a similar two-step workflow is used:
☆ First-line test (widely available): FIB-4
FIB-4 blood test is used, with the following fibrosis risk thresholds:
Low-risk: FIB-4<1.3
Indeterminate: FIB-4 1.3-2.67
High-risk: FIB-4>2.67
☆ Second-line test: elastography and/or ELF
Individuals with indeterminate or high FIB-4 are recommended to undergo secondary risk stratification with liver elastography (e.g., VCTE) and/or the Enhanced Liver Fibrosis (ELF) test. Low-risk patients typically remain in primary care with periodic reassessment to monitor progression.
Figure 8 A generalized risk stratification pathway for MASLD/MASH[20]
☆ Monitoring disease status
Longitudinal assessment of LSM and/or CAP supports routine clinical management and patient counseling. VCTE has become a preferred non-invasive tool for ongoing disease monitoring in MASLD/MASH.
☆ Assessing treatment response
Serial VCTE measurements may also be used during treatment, offering insight into patient response and supporting clinical decision-making. When interpreted in context, these data can help guiding treatment adjustment.
2.3 Diagnostic performance VCTE should not be viewed as a definitive diagnostic replacement for histology. Its performance profile is characterized by strong role-out sensitivity (high negative predictive value, NPV), with more limited rule-in specificity[21].
High sensitivity:
In screening and triage, minimizing false negatives is prioritized so that patients with liver fibrosis are not missed. This favors sensitivity and supports VCTE as a front-line assessment tool.
Compromised specificity:
Measurements could be influenced by factors beyond fibrosis, including obesity, inflammation, and congestion, which may elevate stiffness values and increase false positives. In addition, prioritizing sensitivity increases the number of patients falling into indeterminate or positive ranges, further reducing specificity.
This trade-off is explicitly reflected in guideline dual-cutoff pathways. VCTE and LB are often used in a complementary manner:VCTE enables scalable screening and triage, while LB remains the confirmatory reference when definitive staging or histological results are required, particularly in clinical trials.

3.1 How VCTE is used in trials today
In MASLD/MASH clinical development, VCTE mainly plays two roles, from pre-screening to treatment-response surveillance. Reflecting this application, we reviewed relevant clinical trials (excluding lifestyle/behavioral and supportive-care studies) registered as Phase 2-4 and with statuses of “completed”, “recruiting”, “active not recruiting” or “not yet recruiting”. To the end of 2025, this search identified 87 trials, of which 38 include VCTE, most commonly as an eligibility criterion and/or as a secondary or exploratory endpoints, with only a small minority specifying VCTE-derived measures as primary endpoints[22].
Figure 9 Summary of VCTE (FibroScan®) application in clinical trials (To 2025)
Two Phase 3 studies are presented as examples to demonstrate how VCTE is implemented in late-stage study design. These studies are chosen solely for clarity in reporting within the scope of this research.
3.2 Case Examples
3.2.1 ESSENCE Trial (NCT04822181)
ESSENCE is a randomized, multicenter, double-blinded, parallel-group Phase 3 trial to evaluate the effect of semaglutide (subcutaneous, 2.4 mg) in MASH patients with fibrosis F2 or F3[23].
☆ Trial endpoints
While the primary endpoints follow FDA-accepted histologic surrogates, the study strengthens its evidence chain by including FibroScan-based changes in liver stiffness and CAP as supportive secondary endpoints.
Figure 10 NCT04822181 clinical trial basic information
Figure 11 NCT04822181 study design
☆ Screening period
The ESSENCE added a pre-qualification step prior to confirmatory LB. This step was designed to enrich the screened population for individuals more likely to meet biopsy-based eligibility criteria[24]. During pre-qualification, participants need to meet at least one of the following criteria:
* LB performed within 180 days before Visit 1 and available for central review
* History of any of the following NITs:
ELF ≥ 9.8, FibroScan® ≥ 9.1 kPa, MRE ≥ 3.2 kPa, FAST ≥ 0.67, or biopsy consistent with NASH and presence of fibrosis stage 2 or 3
* Fib-4 ≥ 1.3 measured at Visit 1
In the current dataset, approximately 91% of enrolled participants had at least one positive NIT result concordant with histologically confirmed disease, supporting the effectiveness of this enrichment approach[24]
Figure 12 Assessment timepoints of FibroScan and liver biopsy[23]
Efficacy At Week 72, semaglutide treatment was associated with greater resolution of histological disease activity and a more pronounced reduction in liver stiffness measured by VCTE (VCTE-LSM) compared to control. The VCTE confidence intervals were unadjusted for multiplicity and was interpreted descriptively[25].
Figure 13 Efficacy measured by liver histology and VCTE
VCTE’s Role
* Pre-qualification screening criteria
* Supportive secondary endpoints
3.2.2 MAESTRO-NASH Trial (NCT03900429)
MAESTRO-NASH is multicenter, double-blind, randomized, placebo-controlled Phase 3 trial to evaluate the effect of resmetirom (oral, 80 mg, 100 mg) in MASH patients with fibrosis F1B-F3. This study was designed to support two regulatory approval milestones, accelerated approval based on Week 52 liver histology, and full approval contingent upon Month 54 clinical outcomes[26][27].
☆ Trial endpoints
Two primary endpoints are histology-based. FibroScan-derived measurement changes from baseline are evaluated as part of secondary endpoints.
Figure 14 NCT03900429 clinical trial basic information
Figure 15 NCT03900429 study design
☆
Screening period
Similar to the ESSENCE, a prescreening step was implemented[28]. Key prescreening criteria include:
* No history of significant alcohol consumption.
* ≥3 metabolic risk factors per modified IDF criteria.
* Liver Disease Evidence (Must meet one of the following):
◆ Elevated AST (Women >17 IU; Men >20 IU) AND a FibroScan (within 3 months) showing LSM ≥8.5 kPa & CAP ≥280 dB/m.
◆ A historical liver biopsy (<2 years) confirming fibrosis stage 1B, 2, or 3 with NASH.
Efficacy (VCTE-derived data only)
VCTE data indicated a consistent improvement in liver stiffness with resmetirom at Week 52[27]. The 100 mg treatment group showed the greatest response, with the largest percentage of patients meeting the ≥25% improvement. CAP improvement suggested reductions in steatosis, further indicating a beneficial effect.
Figure 16 Efficacy measured by liver histology and VCTE[27]
VCTE’s Role
* Prescreening criteria
* Secondary endpoint
3.3 A summary
Semaglutide received FDA conditional accelerated approval for MASH on August 15, 2025, based on data obtained from the ESSENCE[29]. In January 2026, CHMP from EMA issued a recommendation supporting conditional marketing authorization, suggesting the European approval may be forthcoming[30].
Currently, VCTE is not considered a substitute for liver biopsy for pivotal decision-making in most programs. Histological measures are still mandatory and form a critical component for regulatory approval[31]. That said, a minority of trials, have emerged to adopt VCTE-derived measurement as primary endpoints. This may point to a growing confidence in the technology and an effort toward less invasive evaluation[22].
Figure 17 Clinical trials with VCTE measurements as primary endpoints[22]
4.1 FDA regulatory framework for biomarker qualification
FDA’s acceptance of Echosens’ LOI represents an initial step within the Biomarker Qualification Program. Qualification requires clarity on both the evidence expected and the process by which that evidence is reviewed. These considerations are articulated in two FDA guidance documents that together frame both the scientific bar and the procedural path for evaluating LSM by VCTE.
4.1.1 Biomarker Qualification: Evidentiary Framework, Guidance for Industry and FDA Staff[33]
ESSENCE is a randomized, multicenter, double-blinded, parallel-group Phase 3 trial to evaluate the effect of semaglutide (subcutaneous, 2.4 mg) in MASH patients with fibrosis F2 or F3[23].
Figure 18 FDA draft guidance on biomarker qualification[33]
This draft guidance clarifies that qualification is context-of-use (COU) specific and organizes evidence expectations into four domains:
1) Needs Assessment
Explains why a biomarker is needed in drug development, its added value relative to existing tools, and how it could improve or accelerate drug development, particularly for diseases with unmet medical needs.
2) COU
Each qualification request should correspond to one clearly defined COU, typically described by:
* Biomarker category (BEST category)
Diagnostic, monitoring, pharmacodynamic/response, predictive, prognostic, safety, or susceptibility/risk biomarker.
* Proposed use in drug development
Include the purpose, trial population or model system, and therapeutic mechanism of action when relevant to the biomarker’s biology and intended utility.
3) Assessment of Benefits and Risks
Developers should present a summary of expected benefits and potential risks, along with proposed risk mitigation strategies. This profile informs whether the overall evidence package is sufficient for qualification.
4) Determining Evidence that is Scientifically Sufficient to Support COU
Evidence required depends on the COU and benefit-risk considerations. Where benefits clearly outweigh risks (or risks can be mitigated), greater uncertainty may be acceptable. Evidence should cover both:
* Clinical validation
* Analytical validation
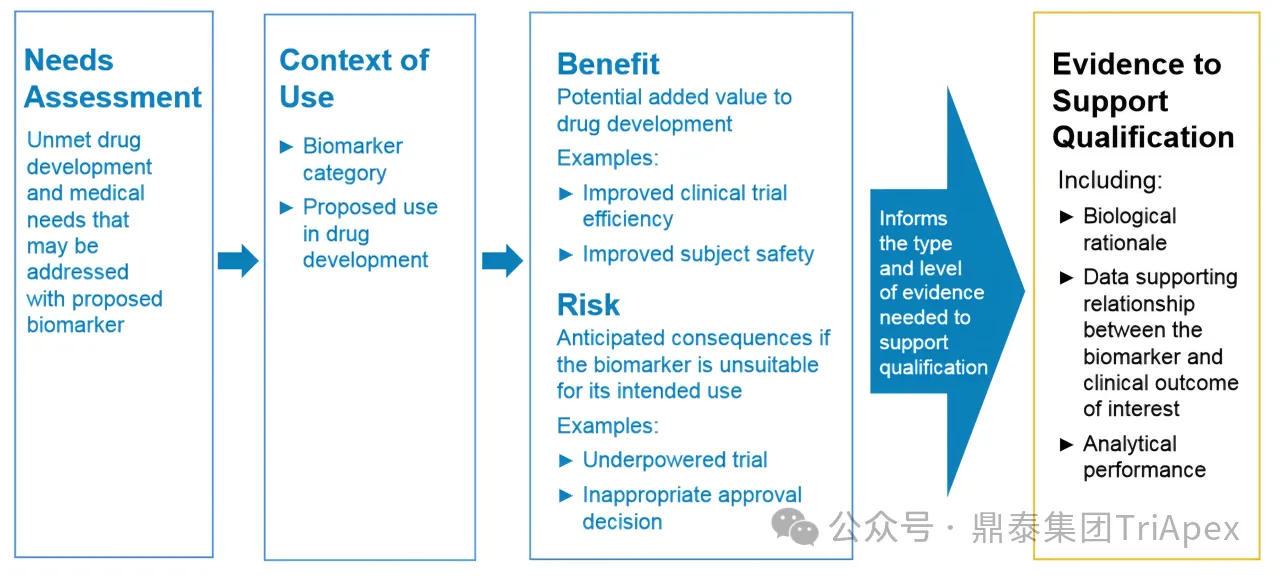
Figure 19 Proposed evidentiary framework by FDA[33]
4.1.2 Qualification Process for Drug Development Tools, Guidance for Industry and FDA Staff[34]
FDA guidance on drug development tool (DDT) qualification process describes an iterative review workflow and duration. FDA issues a decision at each stage:
* Letter of Intent (LOI) – target review within 3 months
* Qualification Plan (QP) – target review within 6 months
* Full Qualification Package (FQP) – target review within 10 months
Figure 20 FDA guidance on qualification process for DDTs[34]
These timelines represent target milestones rather than fixed deadlines and may be subject to adjustment based on the completeness of submissions and the extent of FDA-requestor interaction.
4.2 Potential expansion beyond clinical trials VCTE’s use in animal studies appears to be comparatively less mature and more heterogenous. Much of the published work has focused on feasibility, repeatability, and technical adaptation as prerequisites for broader experimental or veterinary use[35][36][37][38]. This may suggest that the field is still consolidating methodological foundations and clarifying readout interpretation across different disease contexts.
Looking forward, the role of animal elastography may evolve in response to the regulatory and translational emphasis on noninvasive biomarkers. FDA’s recent action may incentivize sponsors to pursue clearer nonclinical-to-clinical continuity. Considerations summarized below offers a preliminary synthesis of factors that may influence how animal VCTE could be applied in future nonclinical studies.
Figure 21 Preliminary considerations for future animal VCTE use
Taken together, current evidence suggests that VCTE in animals can be discussed most conservatively as a supportive translational tool, with potential utilities that may range from disease monitoring to pharmacology evaluation.

The rising burden of MASH creates an urgent need for the accelerated therapeutic development. VCTE, supported by extensive prior use and data, offers a noninvasive approach that could complement histology in MASH trials. It may improve development efficiency by reducing screen failure rates and enabling more frequent assessment than biopsy-based approaches alone. Recent regulatory momentum around VCTE may catalyze further investment into NITs, encouraging the generation of more qualification-grade evidence, thereby accelerating methodological maturation across the broader biomarker ecosystem for MASH.
As an innovative, next-generation CRO, TriApex works to translate evolving scientific and regulatory expectations into study designs and evidence packages. We aim to support more efficient MASH drug development and advance progress in this high-unmet-need field, while looking forward to continued regulatory progress and NIT development in this field.
[1]Younossi ZM, Kalligeros M, Henry L. Epidemiology of metabolic dysfunction-associated steatotic liver disease. Clin Mol Hepatol. 2025;31(Suppl):S32-S50.
[2]Targher G, Valenti L, Byrne CD. Metabolic Dysfunction-Associated Steatotic Liver Disease. N Engl J Med. 2025;393(7):683-698.
[3]Tantu MT, Farhana FZ, Haque F, et al. Pathophysiology, noninvasive diagnostics and emerging personalized treatments for metabolic associated liver diseases. npj Gut Liver. 2025;2(18).
[4]Eslam M, Sanyal AJ, George J; International Consensus Panel. MAFLD: A Consensus-Driven Proposed Nomenclature for Metabolic Associated Fatty Liver Disease. Gastroenterology. 2020;158(7):1999-2014.e1.
[5]European Association for the Study of the Liver (EASL); European Association for the Study of Diabetes (EASD); European Association for the Study of Obesity (EASO). EASL-EASD-EASO Clinical Practice Guidelines on the management of metabolic dysfunction-associated steatotic liver disease (MASLD). J Hepatol. 2024;81(3):492-542.
[6]https://www.fda.gov/drugs/development-resources/table-surrogate-endpoints-were-basis-drug-approval-or-licensure
[7]https://www.fairhealthconsumer.org/medical/search/Liver%20biopsy
[8]Fichez J, Mouillot T, Vonghia L, et al. Non-invasive tests for fibrotic MASH for reducing screen failure in therapeutic trials. JHEP Rep. 2025;7(4):101351.
[9]Anania FA, Dimick-Santos L, Mehta R, Toerner J, Beitz J. Nonalcoholic Steatohepatitis: Current Thinking From the Division of Hepatology and Nutrition at the Food and Drug Administration. Hepatology. 2021;73(5):2023-2027.
[10]FDA. Noncirrhotic Nonalcoholic Steatohepatitis With Liver Fibrosis: Developing Drugs for Treatment Guidance for Industry. 2018
[11]https://www.echosens.com/
[12]Yoneda M, Yoneda M, Fujita K, et al. Transient elastography in patients with non-alcoholic fatty liver disease (NAFLD). Gut. 2007;56(9):1330-1331.
[13]Ozercan AM, Ozkan H. Vibration-controlled Transient Elastography in NAFLD: Review Study. Euroasian J Hepatogastroenterol. 2022;12(Suppl 1):S41-S45.
[14]Williams EE, Vuppalanchi R. Role of Vibration-Controlled Transient Elastography in the Evaluation and Management of Metabolic Dysfunction-Associated Steatotic Liver Disease. Curr Hepatology Rep. 2024;23: 355–363.
[15]Newsome PN, Sasso M, Deeks JJ, et al. FibroScan-AST (FAST) score for the non-invasive identification of patients with non-alcoholic steatohepatitis with significant activity and fibrosis: a prospective derivation and global validation study. Lancet Gastroenterol Hepatol. 2020;5(4):362-373.
[16]Sanyal AJ, Foucquier J, Younossi ZM, et al. Enhanced diagnosis of advanced fibrosis and cirrhosis in individuals with NAFLD using FibroScan-based Agile scores. J Hepatol. 2023;78(2):247-259.
[17]Rinella ME, Neuschwander-Tetri BA, Siddiqui MS, et al. AASLD Practice Guidance on the clinical assessment and management of nonalcoholic fatty liver disease. Hepatology. 2023;77(5):1797-1835.
[18]Cusi K, Isaacs S, Barb D, et al. American Association of Clinical Endocrinology Clinical Practice Guideline for the Diagnosis and Management of Nonalcoholic Fatty Liver Disease in Primary Care and Endocrinology Clinical Settings: Co-Sponsored by the American Association for the Study of Liver Diseases (AASLD). Endocr Pract. 2022;28(5):528-562.
[19]Kanwal F, Shubrook JH, Adams LA, et al. Clinical Care Pathway for the Risk Stratification and Management of Patients With Nonalcoholic Fatty Liver Disease. Gastroenterology. 2021;161(5):1657-1669.
[20]Allen AM, Charlton M, Cusi K, et al. Guideline-based management of metabolic dysfunction-associated steatotic liver disease in the primary care setting. Postgrad Med. 2024;136(3):229-245.
[21]Fang JM, Cheng J, Chang MF, Ahn J, Westerhoff M. Transient elastography versus liver biopsy: discordance in evaluations for fibrosis and steatosis from a pathology standpoint. Mod Pathol. 2021;34(10):1955-1962.
[22]https://data.pharmacodia.com/
[23]NCT04822181 protocol
[24]Newsome PN, Sanyal AJ, Engebretsen KA, et al. Semaglutide 2.4 mg in Participants With Metabolic Dysfunction-Associated Steatohepatitis: Baseline Characteristics and Design of the Phase 3 ESSENCE Trial. Aliment Pharmacol Ther. 2024;60(11-12):1525-1533.
[25]Sanyal AJ, Newsome PN, Kliers I, et al. Phase 3 Trial of Semaglutide in Metabolic Dysfunction-Associated Steatohepatitis. N Engl J Med. 2025;392(21):2089-2099.
[26]NCT03900429 protocol
[27]Harrison SA, Bedossa P, Guy CD, et al. A Phase 3, Randomized, Controlled Trial of Resmetirom in NASH with Liver Fibrosis. N Engl J Med. 2024;390(6):497-509.
[28]Harrison SA, Ratziu V, Anstee QM, et al. Design of the phase 3 MAESTRO clinical program to evaluate resmetirom for the treatment of nonalcoholic steatohepatitis. Aliment Pharmacol Ther. 2024;59(1):51-63.
[29]https://www.fda.gov/drugs/news-events-human-drugs/fda-approves-treatment-serious-liver-disease-known-mash
[30]https://www.ema.europa.eu/en/news/meeting-highlights-committee-medicinal-products-human-use-chmp-26-29-january-2026
[31]Davison BA, Harrison SA, Cotter G, et al. Suboptimal reliability of liver biopsy evaluation has implications for randomized clinical trials. J Hepatol. 2020;73(6):1322-1332.
[32]Xanthakos SA, Ibrahim SH, Adams K, et al. AASLD Practice Statement on the evaluation and management of metabolic dysfunction-associated steatotic liver disease in children. Hepatology. 2025;82(5):1352-1394.
[33]FDA. Biomarker Qualification: Evidentiary Framework Guidance for Industry and FDA Staff. 2018.
[34]FDA. Qualification Process for Drug Development Tools Guidance for Industry and FDA Staff. 2020.
[35]Elshaarawy O, Alquzi SHM, Piecha F, Rausch V, Mueller J, Mueller S. Pharmacological Modulation of Liver and Spleen Stiffness in a Cirrhotic Rat Model. Hepat Med. 2025;17:91-104.
[36]Rivero-Juárez A, Morgaz J, Camacho A, et al. Liver stiffness using transient elastography is applicable to canines for hepatic disease models. PLoS One. 2012;7(7):e41557.
[37]Ono M, Imamura Y, Irie Y, Aritsune M, Nishioka S, Horikita T. Transient elastography: a novel, non-invasive method for the evaluation of liver stiffness and controlled attenuation parameter in cows. J Vet Med Sci. 2020;82(5):559-565.
[38]Pyka D, Noszczyk-Nowak A, Krawiec K, Świetlik T, Opieliński KJ. Innovative Elastography Measuring Cap for Ex Vivo Liver Condition Assessment: Numerical and Preclinical Studies in a Porcine Model. Livers. 2025; 5(1):3.
免责声明:本文来自鼎泰集团内容团队,已获得转发授权。本文仅作信息交流而非商业盈利之目的,内容仅供分享学习。
 已关闭
已关闭
固定模块































